Real-World Research (RWR) – Simple Studies that Present Unique Challenges and Deliver Important Outcomes
What is Real-World Research?
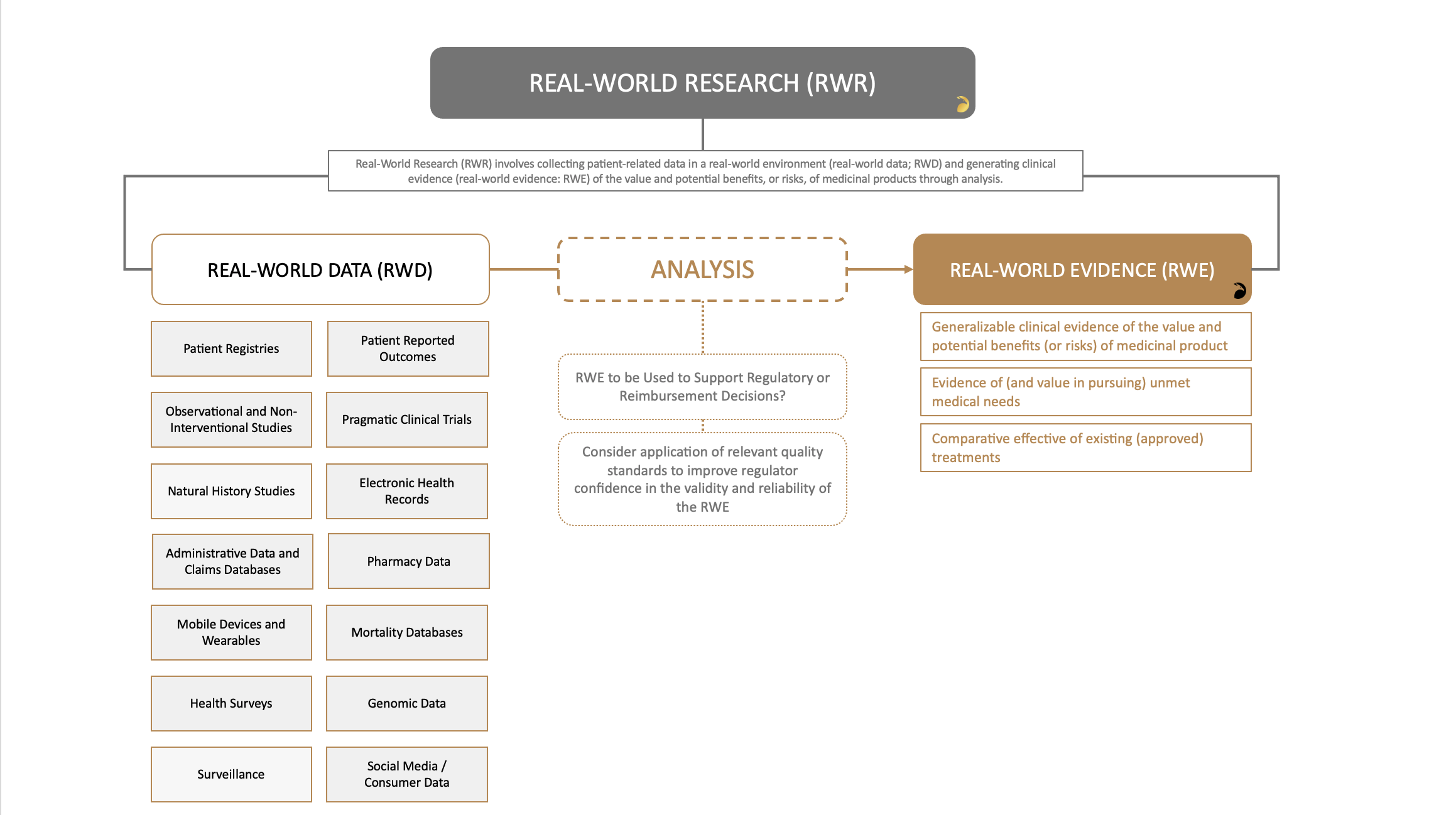
Real-World Research (RWR) involves collecting patient-related data in a real-world environment (real-world data – RWD) and generating clinical evidence (real-world evidence – RWE) of the value and potential benefits, or risks, of medicinal products through analysis.
Examples of real-world research include non-interventional studies, observational studies, registries and pragmatic clinical trials.
Find out more about how real-world analysis can help your business by getting in touch with the global RWR team at Phoenix today.
Why is Real-World Research Important?
Real-world research, inclusive of pragmatic clinical trials, can be used to generate real-world evidence (RWE) to support drug (and vaccine) development, registration, and reimbursement, and can help to address the ‘limitations’ of traditional clinical trials by providing information about what actually happens in the ‘real world’ (e.g., comparative effectiveness of a drug, lack of effectiveness of a vaccine, polypharmacy safety signals, rare adverse events) versus what we thought would happen based on the experimental results from traditional clinical trials. With years of experience advising clients from Chicago, Princeton, San Francisco and globally, we are perfectly placed to help you approach your real-world evidence study with confidence.
RWE Can be Used to Support Drug Development
Real-world evidence (RWE) can support drug development through a variety of ways, covering pre-marketing clinical development and post-marketing evaluation (see Figure 1). Scenarios where real-world evidence can support drug development and regulatory decisions:
Guiding clinical trial design
Identify the target population
Treatment of rare diseases
Revision of indications or drug combination labeling
Post-marketing evaluation and surveillance
RWD Can be Used to Improve the Efficiency of Clinical Trials
Real-world data (RWD) can be used to improve the efficiency of clinical trials, even if not used to generate RWE regarding product effectiveness. For example, RWD can help with:
Generating hypotheses for testing in randomized controlled trials
Identifying drug development tools (including biomarker identification)
Assessing trial feasibility by examining the impact of planned inclusion/exclusion criteria in the relevant population, both within a geographical area or at a particular trial site
Informing prior probability distributions in Bayesian statistical models
Identifying prognostic indicators or patient baseline characteristics for enrichment or stratification
Assembling geographically distributed research cohorts (e.g., in drug development for rare diseases or targeted therapeutics)
[Source: FDA – Framework for Real World Evidence Program, Dec 2018 (USA)]
Figure 1 – Source of Real-World Data (RWD) and Uses of Real-World Evidence (RWE)
RWD ‘Cheat Sheet’ of Quality Factors needed to Ensure ‘Acceptability’ of RWE to Regulators
Key factors to determine whether the quality of real-world data can support drug development include (but are not limited to):
A clear process and qualified personnel for data collection
Common defining framework, i.e., a common data dictionary, is used
Common time frame for key data points collection is followed
Study plan, protocol and/or analysis plan related to the collection of real-world data have been established
Technical approach used for data element capture, including integration of data from various sources, data records of drug use, links to claims data etc., is adequate
Patient recruitment minimizes bias and reflects the true target population
Data entry and transfer are useable and timely
Adequate and necessary patient protection measures such as patient privacy protection and regulatory compliance with informed consent are in place
We Need to Approach Real-World Research with a Different Mindset to Traditional Clinical Trials
Why?
The majority of real-world research study designs are not clinical trials. This is where the regulatory and operational complications start.
OK, fair enough…So HOW should we approach Real-World Research?
Start from first principles. Determine which regulatory ‘framework’ fits your research study…is it a clinical trial? If not, what is it? And then consider…are there local nuances that impact whether the study can be run (e.g., biosampling in Germany) or impact the startup requirements (e.g., study classification in Spain)
Let this inform the regulatory requirements and operational considerations as you build your project playbook from study start-up through to close-out
Real-World Evidence vs. Clinical Trials
Improve the efficiency of your clinical trials using our real-world data analysis. Support drug development and regulatory decision and boost your product effectiveness by discovering how the UK’s leading real-world research experts can support your mission. Having worked with a wide range of clients in San Francisco, Chicago, Princeton, and further afield, we have developed a reputation as one of the world’s leading real-world research and analysis advisors through our hard work, transparency, and dedication to customer satisfaction.
Find out how our team of real-world research advisors can help you ensure acceptability, maintain compliance, and move forward.